Abstract
Multiple myeloma is a post-germinal center B-cell neoplasm, characterized by the proliferation of malignant bone marrow plasma cells, whose survival and proliferation is sustained by growth factors and cytokines present in the bone marrow microenvironment. Among them, IL-6 triggers the signal downstream of its receptor, leading to the activation of the JAK/STAT pathway. The atypical GTPase RhoU lays downstream of STAT3 transcription factor and could be responsible for mediating its effects on cytoskeleton dynamics. Here we demonstrate that RHOU is heterogeneously expressed in primary multiple myeloma cells and significantly modulated with disease progression. At the mRNA level, RHOU expression in myeloma patients correlated with the expression of STAT3 and its targets MIR21 and SOCS3. Also, IL-6 stimulation of human myeloma cell lines up-regulated RHOU through STAT3 activation. On the other hand, RhoU silencing led to a decrease in cell migration with the accumulation of actin stress fibers, together with a decrease in cyclin D2 expression and in cell cycle progression. Furthermore, we found that even though lenalidomide positively regulated RhoU expression leading to higher cell migration rates, it actually led to cell cycle arrest probably through a p21 dependent mechanism. Lenalidomide treatment in combination with RhoU silencing determined a loss of cytoskeletal organization inhibiting cell migration, and a further increase in the percentage of cells in a resting phase. These results unravel a role for RhoU not only in regulating the migratory features of malignant plasma cells, but also in controlling cell cycle progression.
Introduction
Multiple myeloma (MM) is a post-Germinal Center cancer characterized by a multifocal proliferation of clonal, long-lived plasma cells (PCs) within the bone marrow (BM)1. This multistep malignancy is preceded by an age-progressive premalignant condition called monoclonal gammopathy of undetermined significance (MGUS)1–3. Some patients pass through a phase called smoldering myeloma (sMM), in which some of the diagnostic criteria for MM are met but there are no clinical manifestations2. In early stages, MM cells like normal long-lived PCs are highly dependent on the BM microenvironment that activates multiple pathways, protecting these cells from apoptosis4. IL-6, primarily produced by BM stromal cells (BMSCs), is the best characterized MM growth factor and is highly responsible for cell homing, seeding, proliferation, and survival through the activation of the JAK/STAT pathway2,4.
The Rho family of small guanosine triphosphatases (GTPases) forms part of the Ras super-family. These GTPases share a common biochemical mechanism, acting as molecular switches to transduce the signal downstream to their effectors5. To note, the Ras family has been proven to profoundly influence cell growth and activating mutations of Ras are associated with cancer6. In contrast, Rho GTPases are hardly ever found mutated but often display altered activity in malignant cells when compared to healthy counterparts7. Rho GTPases are potent regulators of cytoskeleton dynamics and of the actin filament system, thereby affecting the morphologic and migratory properties of cells8. Due to their important roles in controlling these cellular processes, deregulated Rho GTPases could be at the basis of many tumorigenic events.
The RhoU/V sub-family is particularly interesting due to its unique domain organization. Both members of this family, RhoU and RhoV, have an N-terminal proline-rich domain that is not present in any other Rho GTPase and that enables them to permanently bind to their effectors7,9. RhoU has no detectable GTPase activity but its very high intrinsic guanine nucleotide exchange activity is likely to ensure that the protein is predominantly in the GTP-loaded conformation10. It is encoded by the RHOU gene at 1q42.13 and its expression is mainly controlled at the RNA level downstream of Wnt-1 and STAT3 activation and it might mediate the effects of these signaling pathways in regulating cell morphology, cytoskeletal organization, and proliferation11. Also, different levels of this GTPase might lead to diverse outcomes in cell morphology. It is known that during epithelial-mesenchymal transition of neural crest cells, high levels of RhoU influence cell polarity and migration while low levels are required for cell adhesion12. While typical Rho proteins, such as Cdc42 and Rac1 that share significant sequence homology with RhoU, have an established role in cancer, very little is known about RhoU in tumorigenesis in particular in hematologic malignancies7. Since RhoU can alter cell adhesion, actin dynamics, and cell motility, we aimed at testing if this protein could mediate these cellular features in myeloma cells and if changes in its expression, and thus activity, might lead to BM niches remodeling.
Materials and methods
Patient samples and healthy donors
PCs were purified from BM samples using CD138 immunomagnetic microbeads (MidiMACS system, Miltenyi Biotec, Auburn, CA) and the purity of the positively selected PCs was >90% in all cases. Gene expression profiles (GEP) were investigated in a panel of 268 patients included in two different datasets and representative of all the major forms of PC dyscrasia: a proprietary dataset at NCBI Gene Expression Omnibus repository (accession #GSE66293) previously profiled by us (4 normal controls and 129 MM patients)13,14; and a publicly available data set including five normal controls, 20 MGUS, 33 SMM, and 41 MM patients (accession #GSE47552)15. The cohort consists of newly-diagnosed patients. The proprietary 129 MM tumors (accession #GSE66293) employed for the study were representative of the major molecular characteristics of the disease and stratified according the TC classification16,17. Deletions of 17p13, 13q14 and gain of 1q were also evaluated by FISH18. Procedures followed the rules indicated in the declaration of Helsinki. The internal Institutional Board approved the use of human material.
Cell lines
INA-6 cell line was a kind gift from Dr. Martin Gramatzki (University of Kiel, Germany). U266, H929, RPMI-8226, and HS-5 were purchased from ATCC-LGC Standards (Milan, Italy). SaMMi cell line was generated in our laboratory19. Cell cultures were kept under controlled atmosphere at 37 °C in the presence of 5% CO2 and were periodically checked for Mycoplasma contamination.
SiRNA transfection
U266 cells were transfected with Amaxa Nucleofector® Kit C (Lonza, USA) and 100 pmol of RhoU siRNA (Dharmacon ON-TARGETplus RHOU siRNA, Euroclone, Italy) or 100 pmol of Scrambled (Dharmacon ON-TARGETplus non-targeting siRNA, Euroclone, Italy) according to the manufacturer’s instruction.
Chemicals
2 × 106 cells were cultured in 2 ml of favorite medium, stimulated with 10 ng of IL-6 (ImmunoTools, Germany) and samples were collected at different time points. Cell lines that are usually cultured in medium supplemented with IL-6 were starved from this cytokine for 12 h previous to stimulus.
Stattic (Selleckchem, USA) was employed for 5 h at different doses to inhibit STAT3 and then IL-6 was added or not to the cells for 1 h. Cells were starved of IL-6 only during the 6 h of treatment.
Lenalidomide (Selleckchem, USA) was used at different doses for 4 or 24 h.
Transwell migration assay
5μmTranswell® Permeable Supports on 24 well plates (Corning, USA) were used according to manufacturer instructions. Six hundred microliter non-supplemented medium + 0.1%BSA were added to the bottom of multi-well plate and 50 μL of the same mix were added on top of the transwell insert. 4 × 105 cells were washed with HBSS buffer and then resuspended in 50 μL of recommended medium + 0.1%BSA and added to the top well insert. Plates were incubated for 20 min and then IL-6 stimulus (10 ng) or MSC culture medium (from 24 h MSC culture) was carefully added to the bottom well. IL-6 or MSC culture medium was not added to control wells. Plates were incubator for 6 h before collection.
Immunohistochemistry (IHC)
IHC was performed on 4 μm-thick formalin-fixed, paraffin-embedded sections of BM biopsies of 1 healthy and 8 MGUS using anti-RhoU (HPA049592, Sigma-Aldrich, USA) and anti-IRF4 (HPA002038, Sigma-Aldrich, USA) monoclonal primary antibodies. Fifteen-miilimeter biopsies were stained with anti-RhoU and zones with a clearly abundant plasmacytosis were used in evaluating RhoU positivity. All sections were processed using the sensitive Bond Polymer Refine Detection kit, a biotin-free, polymeric horseradish peroxidase–linker antibody conjugate system, in an automated immunostainer (Bond maX, Menarini, Italy). Appropriate positive and negative controls were run concurrently. RhoU immunostain was semiquantitatively scored in a four-tiered scale, as follows: score 0 = negative staining; score 1 = weak positivity staining; score 2 = moderate positivity staining; score 3 = strong positive staining. Immunohistochemical reactions were independently scored by two investigators (agreement k > 0.8).
Immunofluorescence (IF)
5 × 104 cells were seeded on polylysine-coated glass slides and incubated at 37 °C for 1 h to let them adhere to the polylysine, fixed with formaldehyde 3.7%, permeabilized with Triton 0.1% and blocked with BSA 3%. Samples were then stained with Phalloidin Alexa Fluor 594 (Invitrogen, USA) for 30’, mounted in Vectashield mounting medium with DAPI (4′,6-diamidino-2-phenylindole) (Vector Laboratories, USA) and analyzed using ZEISS LSM700 confocal microscope with 63× magnification objective. Images were analyzed with ImageJ software.
Results
Rho GTPases display altered expression in MM PCs
We have assessed the expression of the Rho GTPase family members in PCs from BM biopsies of MM patients and in normal BM PCs from healthy donors, using the gene expression profiling (GEP) of 129 MM patient samples at diagnosis and four healthy controls included in the proprietary GEO data set GSE66293. We found that Rho GTPase family members are differently expressed in MM PCs when compared to healthy PCs (Fig. 1a). More precisely, 12 (CDC42, RAC1, RAC2, RHOA, RHOBTB1, RHOD, RHOF, RHOG, RHOH, RHOU, RHOV, and RND1) out of the 21 GTPases analyzed, were expressed at significantly lower levels in MM when compared to healthy controls (p < 0.05).
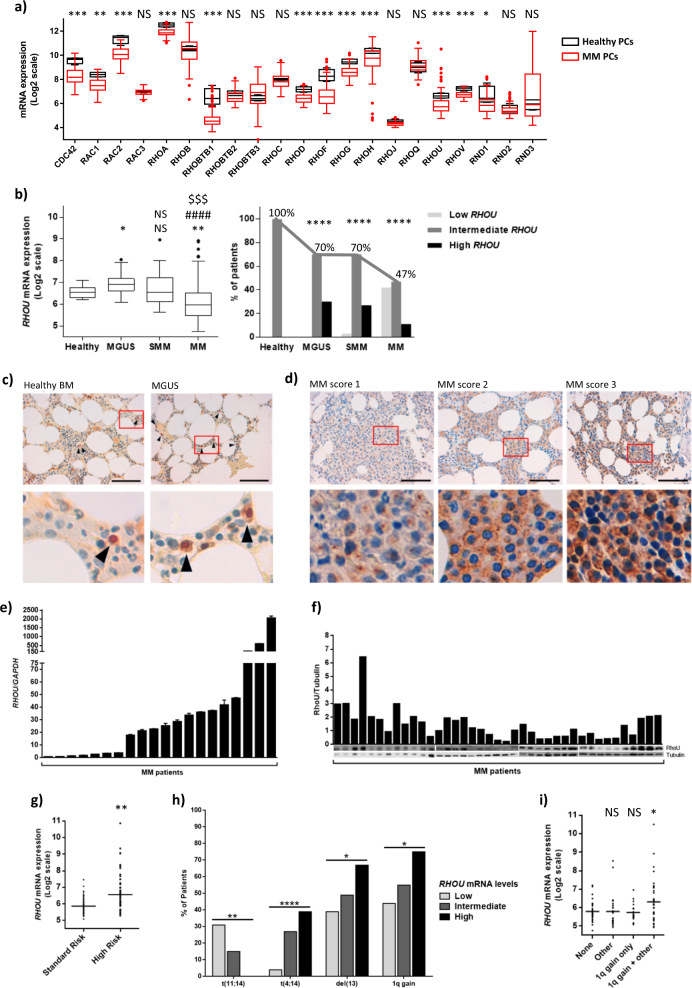
Fig. 1. RhoU is heterogeneously expressed in MM.
a Rho GTPase family members’ expression in healthy and MM PCs. Box plot showing mean ± SD of healthy PCs from four donors (red) and mean ± SD of MM PCs from 129 MM patients at diagnosis (black). In the x axis are all the members of the Rho GTPase family in alphabetical order. Student’s t test *p < 0.05; **p < 0.01; ***p < 0.001 when compared to healthy PCs. b left: RHOU expression in the different steps of MM progression. Box plot showing mean ± SD of healthy PCs from nine donors, MGUS PCs from 20 patients, sMM PCs from 33 patients and MM PCs from 170 patients at diagnosis. Student’s t test: NS non significant; *p < 0.05; **p < 0.01; ***p < 0.001; ****p < 0.0001 (*compared to healthy; #compared to MGUS; $compared to SMM). right: Bar chart showing the percentage of patients for each step of the disease that fall in the diverse groups of RHOU expression. Patients were divided in three groups with low (<mean−2 SD of healthy controls), intermediate, and high (>mean + 2 SD of healthy controls) RHOU expression. Chi square test of patients’ distribution: ****p < 0.0001 when compared to healthy. c RhoU expression in BM biopsies of a healthy donor and a representative MGUS patient. PCs in the samples are indicated with black arrows. Nucleuses of PCs are positive for IRF4 staining and are therefore red. d IHC staining showing low (MM score 1), intermediate (MM score 2) and high (MM score 3) positivity for RhoU staining in BM biopsies from representative MM patients. c and d Original magnification x60; inset with x5 digital zoom; scale bar = 100 μm. e Bar charts showing qRT-PCR RHOU/GAPDH ratio values in PCs purified from 20 MM patient’s biopsies. Mean ± SD of technical triplicates is shown for each samples. f Immunoblot and densitometry of RhoU/Tubulin in PC lysates from 39 MM patient’s biopsies. g Aligned dot plot with RHOU expression in patients that fall in the standard risk (TC1 and TC2) and high risk (TC3, TC4, and TC5) groups. Mean expression in each group is marked with a bar. p value was calculated by Student’s t test. h Bar chart representing the percentage of patients from each of the three groups with low (<mean—2 SD of healthy controls), intermediate, and high (>mean + 2 SD of healthy controls) RHOU expression that exhibit a particular genetic alteration. Student’s t test for trend *p < 0.05; **p < 0.01; ****p < 0.0001. i Aligned dot plot with RHOU expression in patients that don’t present any unfavorable cytogenetic alterations (none), that present unfavorable cytogenetic alterations but not 1q gain (other), that present only 1q gain, and that present 1q gain combined with other unfavorable cytogenetic alterations. Student’s t test: NS non significant; *p < 0.05
RhoU is heterogeneously expressed in different steps of MM progression
Next, we combined the data available in two GEP data sets (#GSE66293 and #GSE47552) as previously described13 and found that RHOU expression is significantly and negatively modulated with disease progression, being over-expressed in BM PCs from MGUS patients and down-modulated in BM PCs from most MM patients (Fig. 1b left panel). However, we found a large heterogeneity in RHOU expression, with several patients showing high transcript levels even in late stages. Therefore, we divided patients in three groups according to low (<mean—2 SD of healthy controls), intermediate (=mean ± 2 SD of healthy controls), and high (>mean + 2 SD of healthy controls) expression. Along with disease progression there was an evident decrease in the percentage of patients that fell in the group with intermediate RHOU expression (Fig. 1b right panel) and an increase in heterogeneity. In MM in particular, there were three distinct groups of patients: 42% with low, 47% with intermediate, and 11% with high RHOU expression.
To further examine RhoU expression we performed IHC analysis in an independent panel of patients including 8 MGUS and 15 active MM. All MGUS patients’ PCs showed a high intensity cytoplasmic staining for RhoU when compared to healthy PCs (Fig. 1c). Also, PCs from all MM patients showed positivity for RhoU staining and were given a score based on the intensity. More precisely 20% showed weak (score 1), 40% moderate (score 2), and 40% high RhoU (score 3) positivity, consistent with the heterogeneous levels of RHOU mRNA described above (Fig. 1d). Cells with different scores seem to also have a diverse localization of RhoU: in MM cells with score 1 RhoU had a dot-like localization in proximity of the nucleus, which could represent the actin microtubule-organizing center, whereas cells that scored 2 or 3 showed a granular cytoplasmic and membrane localization. The heterogeneous levels of RhoU in MM patients were also validated by qRT-PCR (Fig. 1e) and Western Blot (Fig. 1f).
Next, we evaluated RHOU levels according to the major molecular groups of MM patients based on the TC classification as previously described17. Patients were further divided in two groups: standard risk patients that fall into TC1 and TC2 groups (n = 64) and characterized by t(11,14) (TC1) and hyperdiploidy (TC2) leading to cyclin D1 expression, and high risk patients that comprise TC3, TC4, and TC5 groups (n = 65) characterized by cyclin D2 expression alone (TC3) and presence of t(4;14) (TC4) or MAF translocations (TC5). The results show that patients in the high risk group have significantly higher levels of RHOU expression (Fig. 1g), compared to TC1/TC2 patients. Furthermore, patients with high RHOU expression have a higher frequency of 1q gain, del(13), and t(4;14) (Fig. 1h). Finally, since RHOU is located at 1q42 we have divided the patients in four groups, without unfavorable cytogenetic alterations (none), with unfavorable cytogenetic alterations but without 1q gain (other), with 1q gain only, and with 1q gain plus unfavorable cytogenetic alterations, discovering that 1q gain alone is not enough to cause a significant increase in RHOU expression (Fig. 1i).
To better understand if RHOU expression could correlate with patient survival we have used two datasets: one with expression in untreated patients and overall survival after Total Therapy 2 (TT2) and Total Therapy 3 (TT3) regimens (#GSE2658)20–22, and another with expression after several lines of therapy and overall survival after Total Therapy 6 (TT6) (#GSE57317)23. We have divided the patients from each dataset in RHOU expression quartiles and compared the survival curves of the first and fourth quartiles. In TT2 or TT3 regimens there is a 50% higher risk of death (hazard ratio = 1.50) for the patients in the 4th quartile when compared to patients in the I quartile (Supplementary Figure S1a and b). In TT6 regimen the risk of death increases to 612% (hazard ratio = 7.12) for patients in the IV quartile when compared to patients in the I quartile (Supplementary Figure S1c and d). These data suggest a positive correlation between high RHOU levels and worse prognosis also after anti-myeloma therapies. Whether or not its association with poor outcomes can be linked to the combination of 1q gain and other unfavorable cytogenetic alterations remains to be investigated.
Patients with high and low RHOU expression have a different GEP
To gain insights into the role of RHOU in MM, we focused on our proprietary MM group (#GSE66293) due to the absence of any bias related to sample molecular characteristics, and looked for the gene expression signature associated with RHOU expression. Specifically, we ranked patients in four classes based on RHOU expression level. Next, by comparing the lowest (I quartile) with the highest (IV quartile) RHOU expressing patients, we found 557 genes differentially expressed between the two groups that appeared to be gradually modulated in the four quartiles (Supplementary Table S1). Functional enrichment analysis of the 557 deregulated genes using DAVID Bioinformatics Resources 6.8 evidenced 12 significantly annotation clusters (ES > 1.3) and six significantly enriched pathways, among which cell cycle and DNA repair (Supplementary Table S2). In addition, to confirm the correlation between RHOU and these pathways, we used the Gene Set Enrichment Analysis (GSEA) to identify a priori defined sets of genes showing concordant modulation between patients with low and high RHOU expression level. Notably, the cell cycle control and DNA repair gene sets were found highly enriched in MM group expressing lower (I quartile) RHOU levels (Fig. 2a-c).
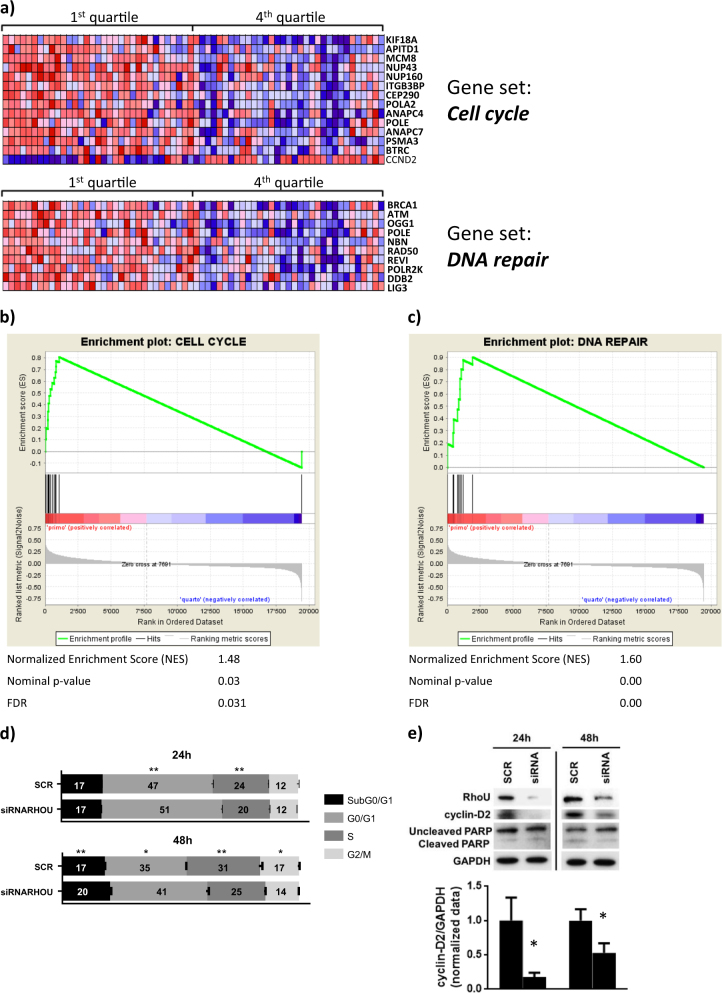
Fig. 2. Patients with high and low RHOU expression have different gene expression profiles.
a Heat map of genes contributing to the core enrichment in the “Cell Cycle” and “DNA Repair” gene sets detected by GSEA analyzing significantly up-regulated genes (p-value ≤ 0.05) in RHOU quartile I versus IV (indicated in bold). Enrichment plots for b “Cell Cycle” and c “DNA Repair” gene sets. The green curves show the enrichment score and reflect the degree to which each gene (black vertical lines) is represented at the bottom of the ranked gene list. d Cell cycle analysis based on PI staining of SCR and siRNA RHOU cells 24 and 48 h after transfection. Data represents mean ± SD of three independent experiments. Student’s t test: *p < 0.05; **p < 0.01. e Representative immunoblots from six samples from two independent experiments of RhoU and cyclin D2 expression 24 and 48 h after siRNA transfection, and densitometry of the mean expression of cyclin D2/GAPDH ± SD
RHOU clustered with 14 genes that regulate the cell cycle and the mitotic process (Table 1). CCND2 was the only gene in the list that positively correlated with RHOU (Pearson’s r = 0.402; p-value = 7.193e-15) suggesting that malignant PCs with high RHOU expression have increased levels of cyclin D2 and a higher replication rate. All the other genes responsible for cell cycle control and DNA damage response had a negative correlation with RHOU expression (Fig. 2b). RHOU also clustered with 10 DNA damage repair genes in the “DNA Repair” pathway (Table 2). All the ten genes in the list negatively correlate with RHOU expression (Fig. 2c).
Table 1.
Gene set “Cell Cycle” was detected by GSEA as significantly up-regulated in RHOU quartile I versus IV
| GSEA results summary | |
|---|---|
| GeneSet | Cell cycle |
| Genes | ITGB3BP, NUP160, BTRC, POLE, ANAPC4, KIF18A, POLA2, MCM8, APITD1, CCND2, PSMA3, CEP290, ANAPC7, NUP43 |
| Normalized Enrichment Score (NES) | 1.48 |
| Nominal p-value | 0.03 |
| FDR | 0.031 |
ITGB3BP overexpression induces apoptosis in cancer cells24; NUP160, NUP43, KIF18A, BTRC, APITD1, and CEP290 are required for correct mitosis, centrosome dynamics, and chromosome alignment25–30; POLE, POLA2, and MCM8 have extremely important roles in DNA replication and genome stability31–33; ANAPC4 and ANAPC7 have emerging roles in differentiation control, genomic stability and tumor suppression34; CCND2 enhances cell cycle progression and MM proliferation35 and PSMA3 is part of the proteasome complex and essential for protein degradation during cell cycle progression36.
Table 2.
Gene set “DNA Repair” was detected by GSEA as significantly up-regulated in RHOU quartile I versus IV
| GSEA results summary | |
|---|---|
| GeneSet | DNA repair |
| Genes | NBN, REV1, POLR2K, POLE, DDB2, LIG3, OGG1, ATM, RAD50, BRCA1 |
| Normalized Enrichment Score (NES) | 1.60 |
| Nominal p-value | 0.00 |
| FDR | 0.00 |
These genes are extremely important since they encode for proteins essential in a number of cellular pathways that maintain genomic stability, including DNA damage-induced cell cycle checkpoint activation, DNA damage repair, protein ubiquitination, chromatin remodeling, as well as transcriptional regulation and apoptosis37–44.
Strikingly, in U266 cell line, the inhibition of RhoU by siRNA resulted at 24 h in a slight but significant accumulation of cells in the G0/G1 phase and a decrease in replicating S phase cells (Fig. 2d). The results were more evident at 48 h with an increase in both sub G0/G1 and G0/G1 phases and a decrease in both S and G2/M phases. Since G1 to S phase transition is tightly regulated by the expression of cyclin D2, we have analyzed this protein’s expression after RhoU silencing and confirmed a decrease in its levels as early as 24 h after transfection; The decrease in RhoU protein levels at these time points was confirmed by western blotting (Fig. 2e).
RHOU is overexpressed in IL-6-dependent MM cell lines and is driven by the activation of STAT3
To better understand RHOU regulation in MM we have investigated its expression on the GEP data (#GSE66293) and on five MM cell lines available in our laboratory, two IL-6 dependent (INA-6 and SaMMi), one that autocrinally produces IL-6 (U266) and two IL-6 independent (H929 and RPMI-8226). GEP data relative to MM cell lines shows that only INA-6, U266 and UTCM2 (a cell line that also autocrinally produces IL-6) had high RHOU expression levels (Fig. 3a). We have also investigated RHOU expression by qRT-PCR confirming that RPMI-8226 and H929 expressed RHOU at very low levels, while IL-6 dependent/autocrine cell lines over-expressed it (Fig. 3b).
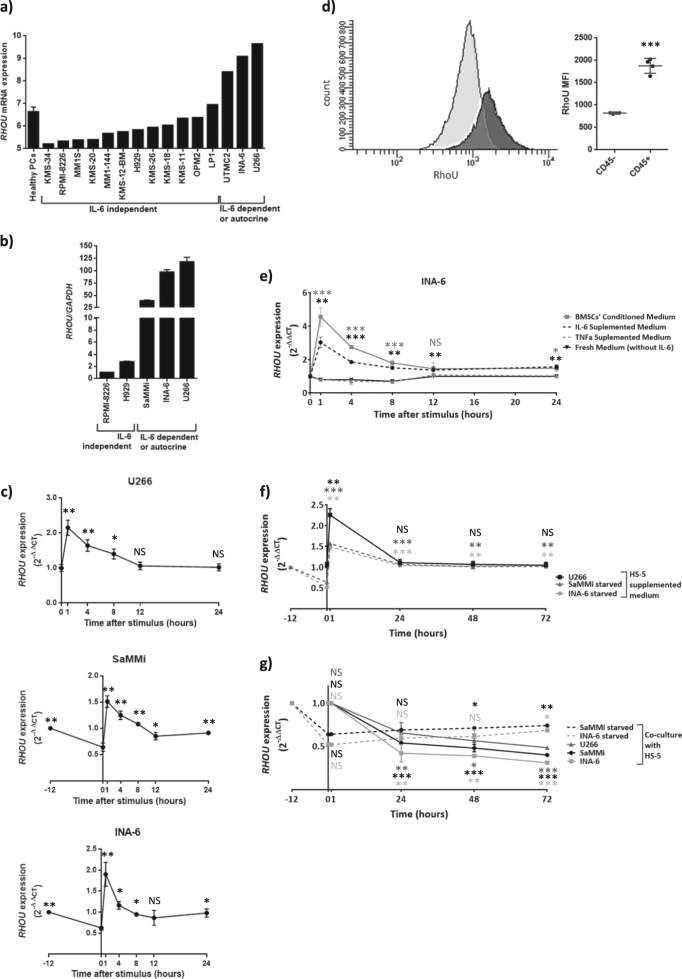
Fig. 3. RHOU expression in MM cell lines relies on IL-6 stimulus.
a Bar chart showing RHOU expression assessed by GEP (#GSE66293) in MM cell lines as compared to healthy PCs. b Bar chart showing RHOU expression in MM cell lines assessed by RT-PCR. Data represents normalized mean ± SD of three independent samples of each cell line. c Time-course of RHOU expression after IL-6 stimulus showing a significant increase in RHOU expression as early as one hour after stimulus; time points before zero correspond to IL-6 starvation period. d Left: representative dot plot showing intracytoplasmic RhoU staining in the CD45+ and CD45− populations of U266 cell line. Right: mean ± SD of four samples mean fluorescence intensity of intracytoplasmic RhoU staining in the CD45+ and CD45− populations of U266 cell line. e Time-course of RHOU expression after the addition of IL-6, TNF-α, BMSCs’ conditioned medium or fresh unsupplemented medium. f Time-course showing the changes in RHOU expression after the addition of HS-5 supplemented medium of starved and not starved cell lines. g Time-course showing the changes in RHOU expression after co-culture with HS-5 of starved and not starved cell lines. c, e, f, and g Data represents mean ± SD of six samples from at least two independent experiments normalized over mean expression at time zero. Student’s t test: NS non significant; *p < 0.05; **p < 0.01; ***p < 0.001, when compared to time zero of untreated
To determine whether IL-6 stimulus could lead to an up-regulation of RHOU expression cells were deprived from this cytokine and subsequently stimulated. Samples were collected at different time points (Fig. 3c). All three IL-6-dependent cell lines up-regulated RHOU as early as 1 h after stimulus. In detail, 1 h after the addition of IL-6, there was an increase in RHOU expression equal to 1.2 times-fold in U266, 1.7 times-fold in SaMMi, and 2.4 times-fold in INA-6. U266 might display the smallest changes due to the fact that this cell line cannot be starved from IL-6 since it autocrinally produces it.
Since CD45+ myeloma cells have increased JAK/STAT activation45,46,, we have also studied the CD45+ and CD45− U266 cell populations to further examine RhoU expression in this context. Intracytoplasmic RhoU staining showed a mean fluorescence intensity (MFI) of 822 and of 1878 in the CD45− and CD45+ cell populations, respectively (Fig. 3d).
To verify if this effect was due to the specific activation of the IL6R/STAT3 pathway, we stimulated cells also with TNF-α, conditioned medium from BMSCs’ culture, or unsupplemented fresh medium (Fig. 3e). The dynamics of RHOU expression after the addition of BMSCs’ conditioned medium or IL-6 stimulus were similar, while TNF-α did not cause any significant changes. Adding derived media from HS-5 stromal cell line showed the same results as IL-6 stimulus (Fig. 3f) and a really slow increase in RHOU expression was obtained by co-culturing IL-6 starved cell lines with HS-5 cells (Fig. 3g). However, the co-culture on HS-5 cells of non-starved cells, with high levels of RhoU, resulted in an actual decrease in RHOU expression (Fig. 3g). These results may support the hypothesis of a contact dependent down-modulation of RHOU after cell adhesion, as previously observed in neural crest cells47.
Interestingly, looking at GEP data from MM patients we found a weak but significant positive correlation between RHOU and STAT3 expression. The analysis of STAT3 targets MIR21 and SOCS3 unraveled however a stronger positive correlation with the expression of RHOU (Fig. 4a).
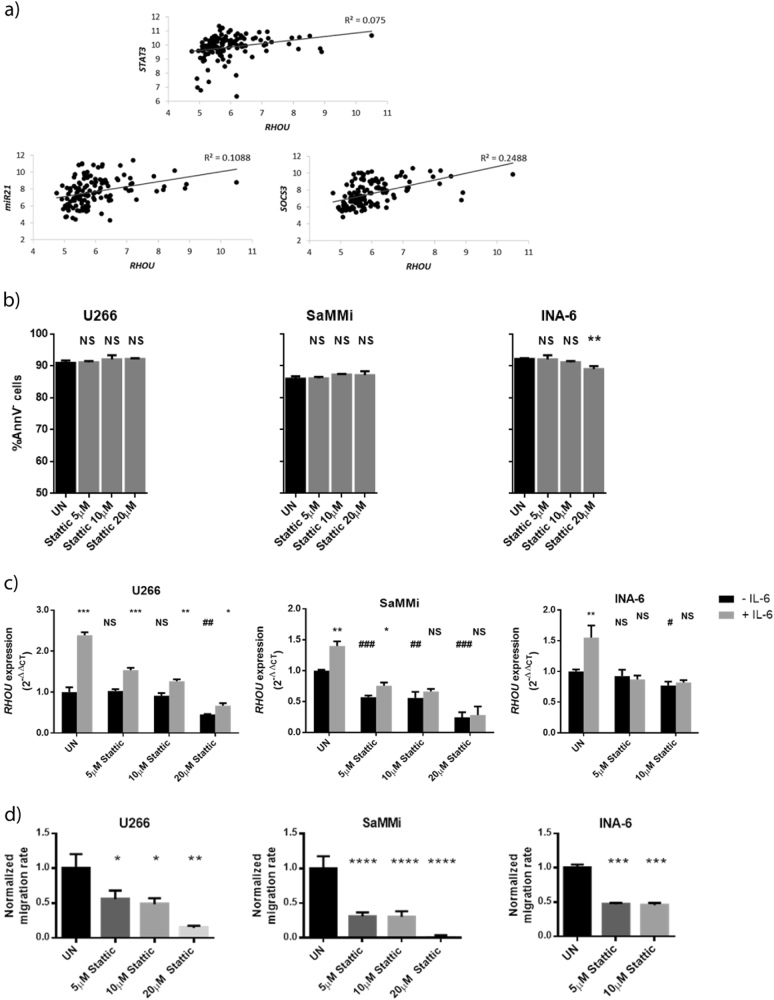
Fig. 4. In MM cell lines RHOU expression lays downstream of STAT3 activation.
a Correlation between RHOU expression and STAT3 expression (Pearson’s r = 0.2739; p-value = 0.0017) or MIR21 expression (Pearson’s r = 0.3299; p-value = 0.0001) or SOCS3 expression (Pearson’s r = 0.4988; p-value = 1.786e-09). b Bar charts showing the percentage of live cells (AnnV−) after treatment with different doses of Stattic. c Bar charts showing RHOU expression after STAT3 inhibition before and after IL-6 stimulus. Black bars represent RHOU expression after 6 h of culture with different concentrations of stattic. Grey bars show RHOU expression after 5 h of stattic treatment + 1 h of combined treatment with IL-6 stimulus. *Compared with same treatment conditions without IL-6 stimulus, #Compared to UN. d Bar charts exhibiting normalized mean cell count ± SD of four samples from at least two independent experiments, after treatment with increasing concentrations of stattic, accessed by migration assay with IL-6, normalized over mean migration of UN. b, c, and d UN untreated; student’s t test: NS non significant; *p < 0.05; **p < 0.01; ***p < 0.001; ****p < 0.0001
Next, to verify that STAT3 was the principal transcriptional regulator of RHOU downstream of the IL-6R, we inhibited STAT3 with stattic, a chemical inhibitor that blocks STAT3 phosphorylation, dimerization, and nuclear transition by specifically binding to its SH2 domain48. Based on annexin V staining, a 6 h treatment with this drug resulted in a slight but significant increase in cell death only at the highest doses for INA-6 cell line (Fig. 4b). Upon non-toxic doses of stattic treatment we observed a dose-dependent decrease in RHOU expression, while cells were less, if not able, to upregulate the GTPase in response to IL-6 stimulus (Fig. 4c). Stattic treatment also led to a dose dependent decrease in cell migration that could rely on RHOU expression (Fig. 4d).
Lenalidomide treatment increases MM cell migration that is overturned by RhoU inhibition
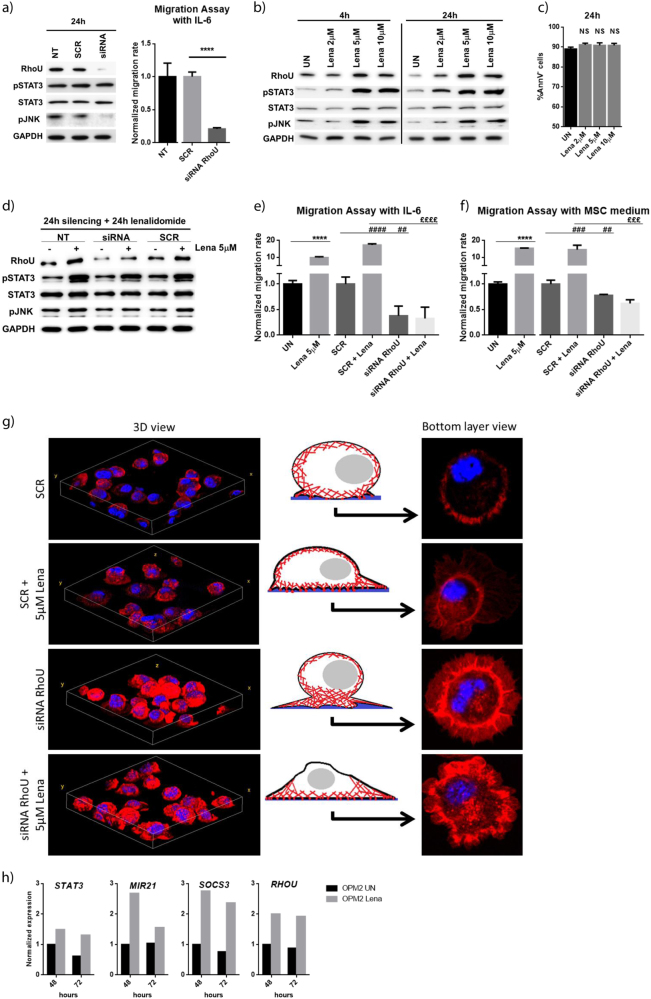
To better assess the effects of RhoU in MM cells we have silenced it by transfecting cells with specific siRNA particles. Scrambled non-targeting siRNA (SCR) was used as control. RhoU silencing led to a clear decrease in JNK activating phosphorylation, but did not seem to have an effect on cell death nor on STAT3 activation (Fig. 5a left panel). JNK1 is a target of RhoU, important for the development of filopodia11. Therefore, an impairment in its activation could explain the decreased migration capability of RhoU-targeting siRNA transfected cells (Fig. 5a right panel). Furthermore, since IMIDs were shown to be able to regulate the activation of classical Rho proteins in human monocytes49, we aimed at studying the effects of lenalidomide on RhoU expression. Immunoblot analysis showed an increase in STAT3 activation after treatment with lenalidomide (Fig. 5b) without a clear effect in viability in U266 (Fig. 5c) and in INA-6 (Supplementary Figure S2a) cell lines. We also observed an increase in RhoU expression and in JNK activating phosphorylation (Fig. 5b and Supplementary Figure S2a) that could result in increased cell mobility. When combining RhoU inhibition with lenalidomide we observed that JNK phosphorylation remained unchanged instead of increasing as happened in UN and SCR conditions (Fig. 5d). When comparing siRNA and SCR transfected cells we observed a significant decrease in migration (Fig. 5e and f). RhoU is thus essential for JNK activation and MM cell migration. Treatment with lenalidomide, that consequently determined higher RhoU and active JNK levels, resulted in a boost in cell migration (Fig. 5e and f; and Supplementary Figure S2b and c). RhoU silencing abolished lenalidomide induced migration (Fig. 5e and f).
Fig. 5. Lenalidomide enhances MM cell migration through the upregulation of RhoU.
a Immunoblot of U266 proteins and bar chart exhibiting normalized mean cell count ± SD of four samples from at least two independent experiments, after 24 h of transfection with SCR or RhoU siRNA. b Immunoblots of U266 proteins after 4 and 24 h of treatment with different doses of lenalidomide. c Bar charts showing the percentage of live cells (AnnV−) after treatment with different doses of lenalidomide for 24 h. d Immunoblot of U266 proteins after 48 h of transfection with RhoU or SCR siRNA, lenalidomide was added in the last 24 h. a, b, and d show representative immunoblots from six samples from at least two independent experiments; UN untreated, NT not transfected; protein expression is showed in the following order: RhoU, phospho-STAT3(Tyr705), STAT3, phospho-JNK(Thr183/Tyr185), and GAPDH. e Bar charts showing normalized mean cell count ± SD of four samples from at least two independent experiments after 48 h of transfection with RhoU or SCR siRNA combined with lenalidomide treatment in the last 24 h accessed by migration assay with IL-6, normalized over mean migration of UN. f Bar charts showing normalized mean cell count ± SD of four samples from at least two independent experiments after 48 h of transfection with RhoU or SCR siRNA combined with lenalidomide treatment in the last 24 h accessed by migration assay with MSC medium, normalized over mean migration of UN. e and f UN untreated *Compared to UN; #compared to SCR; £compared to SCR + Lena, *p < 0.05; **p < 0.01; ***p < 0.001; ****p < 0.0001. g Changes in the actin cytoskeleton after RhoU silencing or SCR transfection combined or not with lenalidomide treatment. DAPI staining shows the nucleus in blue, phalloidin staining in red shows F-actin filaments. Cells were fixed after 48 h siRNA transfection and 24 h of 5 μM lenalidomide treatment. All cells were plated at the same concentration in a six well plate for 48 h to maintain growth conditions. h Expression of STAT3 and its targets MIR21, SOCS3, and RHOU after treatment of OPM2 cell line with lenalidomide for 48 and 72 h (#GSE31421)
We further studied cytoskeleton changes and alterations in cell adhesion by employing IF techniques. Although all cells were able to adhere to polylysine coated glass, the changes in cell morphology were evident. SCR cells displayed a normal round-up morphology (3D view) with smooth edges and few spiky protrusions (bottom layer view); siRNA RhoU cells had bigger lamellipodia-like edges with an accumulation of stress fibers and focal adhesions; lenalidomide treated SCR cells were flattened (3D view) and with multiple filopodial protrusion (bottom layer view), typical of migrating cells in accordance with the migration data above; noteworthy, the combination of RhoU silencing with lenalidomide treatment resulted in a loss of cytoskeleton organization (Fig. 5g).
To deepen the study of STAT3 activation and RhoU overexpression upon lenalidomide treatment, we have analyzed the expression of STAT3 itself and its target genes MIR21, SOCS3 and RHOU in a GEP dataset of OPM2 cell line upon lenalidomide treatment for 48 and 72 h (#GSE31421)50. Upon lenalidomide treatment there is a 50% increase in STAT3 expression, while the increase in the expression of its target genes is even more clear (Fig. 5h).
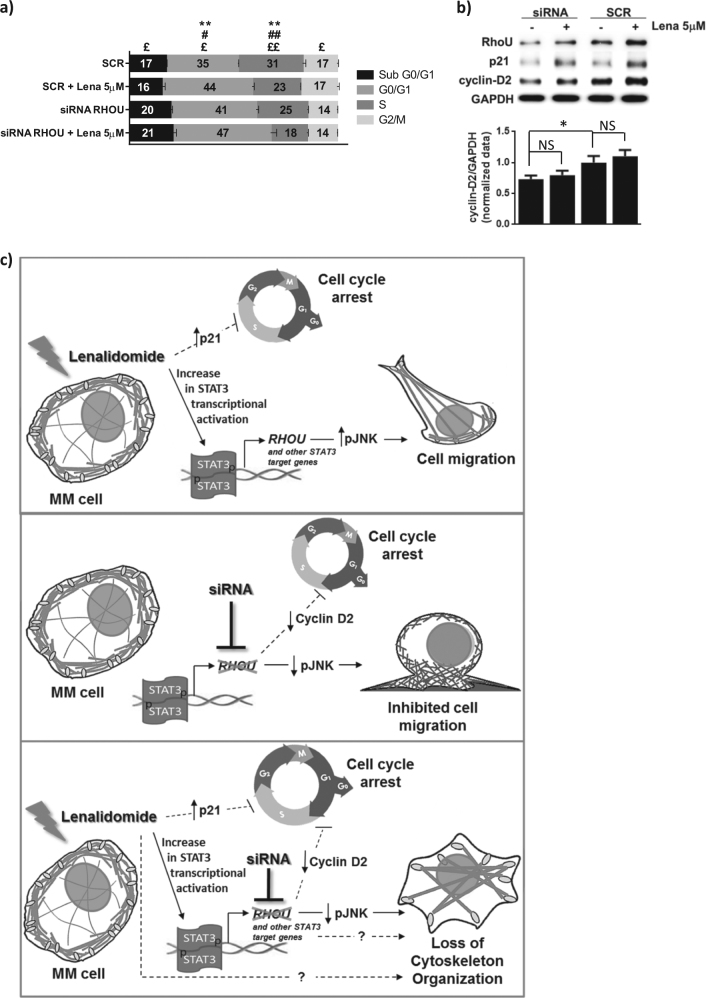
Additionally, since RhoU silencing caused cell cycle arrest we further investigated the changes in cell cycle after lenalidomide treatment or the combination of both. Twenty four-hour treatment with lenalidomide caused a significant increase in G0/G1 phase and a decrease in S phase (Fig. 6a). The combination of both RhoU silencing and lenalidomide led to a further accumulation of cells in the G0/G1 phase (Fig. 6a). We have also analyzed cyclin D2 expression, unraveling that although lenalidomide treatment impaired the transition from G1 to S phases, it did have a significant impact in cyclin D2 expression (Fig. 6b). However, as previously described in literature, lenalidomide treatment caused an increase in p21 expression51 in both conditions that might explain the impairment in cell cycle progression.
Fig. 6. Cell cycle analysis and schematic view of the effect of lenalidomide, RhoU silencing or their combination in MM cells.
a Cell cycle analysis based on PI staining of SCR and siRNA RHOU cells 48 h after transfection with the addition of lenalidomide in the last 24 h. Data represents mean ± SD of three independent experiments. Student’s t test: *SCR compared to SCR + Lena; #siRNA RhoU compared to siRNA RhoU + Lena; £siRNA RhoU + Lena compared to SCR + Lena, *p < 0.05; **p < 0.01. b Representative immunoblots from six samples from at least two independent experiments of RhoU, cyclin D2, and p21 expression after siRNA transfection and lenalidomide treatments, and densitometry of the expression of cyclin D2/GAPDH. c Lenalidomide treatment determines an increase in STAT3 activation and consequently the increased expression of STAT3 target genes including RhoU. This, combined with increased levels of pJNK, results in a reorganization of the cytoskeleton and boosts cell migration. These effects are parallel to cell cycle arrest, through a mechanism that is not explored in this manuscript. RhoU inhibition, on the other hand, results in decreased levels of active JNK leading to decreased cell migration rates and increased lamellipodial protrusions. RhoU silencing also caused MM cell cycle arrest due to a decrease in the expression of cyclin D2. The combination of both RhoU silencing and lenalidomide treatment further inhibits cell cycle progression and overcomes lenalidomide induced migration by decreasing the levels of active JNK causing however a loss of cytoskeleton organization. This cytoskeleton organization loss may be due to the expression of other STAT3 target genes or due to lenalidomide targets not explored in this article
A schematic view of the proposed mechanism is depicted in Fig. 6c.
Discussion
We report here the first evidence of a clear unbalance in the expression of multiple members of the Rho GTPase family in MM PCs when compared to normal PCs. Interestingly, opposite to what was seen in other tumors where there is an over-expression of some of the members of this family7, we have found that more than 50% of Rho GTPases are actually down-modulated in MM PCs when compared to healthy PCs.
Focusing our attention on the RhoU/V family, we have found that RhoU expression is significantly modulated during the different steps of MM progression. MGUS patients showed higher RhoU levels when compared to normal controls, raising the hypothesis that this protein might be important especially in the early stages of MM malignancy. With disease progression and accumulation of malignant PCs in the BM, we observed a decrease in RHOU when compared to MGUS patients. It has been reported that high RhoU levels lead to enhanced cell motility, while low RhoU levels are essential for adhesion47. It is predictable that RhoU is expressed at high levels in the initial steps of the disease when PCs could be more motile since they are competing for BM niches52. With disease progression inside the BM, cells adhere and rely on the microenvironment for their survival, which seems to translate in a cell-contact dependent decrease in RHOU expression, hypothesis supported by the in vitro evidence of a RHOU down-modulation in MM cell lines cultured in contact with HS-5 stromal cells.
Remarkably, MM patients in high risk TC groups have significantly higher levels of RHOU mRNA that positively correlated with the presence of unfavorable cytogenetic alterations; however the sole presence of 1q gain, where RHOU gene is located, was not enough to cause a significant change in its expression. Also, higher levels of RHOU expression correlated with increased CCND2 and decreased cell cycle control and DNA damage repair genes; while RhoU silencing resulted in a decrease in cyclin D2 protein expression and impaired cell cycle progression. Put together, these results suggest that even though most MM patients down-modulate RHOU with disease progression, a high expression of this GTPase in late stages of this malignancy could actually be associated with a worse prognosis. We also found that IL-6 stimulus, important for MM survival, resulted in a STAT3-dependent increase in RHOU expression in cell lines. Likewise, CD45+ U266 cells, that have increased JAK/STAT activation45,46,, exhibited a 2.3 times higher MFI for intracytoplasmic RhoU staining. To note, in MM patients RHOU expression correlated with the expression of STAT3 itself, of MIR21 and of SOCS3, confirming that it might be highly dependent on the activation of the STAT3 cascade, as observed in MM cell lines.
RhoU silencing led to a loss of migration capability probably due to the decrease in active JNK, as previous studies suggested: a constitutively active RhoU mutant was able to increase JNK activation in mouse epithelial cells53 and RhoU depletion in HelaS3 cells inhibited JNK activating phosphorylation consequently impairing filopodium formation, a type of cell protrusion essential for cell migration54. Lastly, we described the effects of IMIDs on the IL-6/STAT3/RhoU/JNK branch. This new generation drugs for MM treatment have been shown to reorganize cells’ cytoskeleton by modulating Rho GTPases in human monocytes49. Indeed, we found that lenalidomide commanded an increased activation of STAT3, augmented RhoU expression and amplified JNK phosphorylation. Parallel to the changes in cytoskeleton conformation after lenalidomide, we also observed a decrease in cell cycle progression, probably through a p21 dependent mechanism, in line with what is already known for this drug51,55,56,. Consistently and opposite to what was observed with RhoU silencing, lenalidomide led to a high increase in cell motility. The combination of RhoU silencing with lenalidomide treatment led to a loss of cytoskeleton organization that translated in a defective migratory capability, and a further increase in cell cycle arrest. These results give insights into a novel mechanism of action of lenalidomide and prove that this drug can somehow stimulate the activation of STAT3 transcription factor enhancing the expression of its target genes including RHOU, leading to cytoskeletal changes in MM PCs. The increased cell motility upon lenalidomide is consistent with the disruption of microenvironment niche previously described57,58, where less adherent cells are more easily targeted by immune cells or combined chemotherapeutics. Nevertheless, whether or not high RhoU levels indicate an increased propensity towards dissemination of malignant PCs, whether RhoU levels during lenalidomide therapy in MM patients are increased, and whether RhoU high-expressing MM could display different responsiveness to lenalidomide remains to be clarified.
Electronic supplementary material
Acknowledgements
We thank patients and their families for donating samples. This work was supported by grants from the Associazione Italiana per la Ricerca sul Cancro (AIRC) to F.P. (IG14481; IG18387), G.S. and A.N., from the Italian Ministry of Education, University and Research (FIRB-Futuro in Ricerca 2008) to F.P. and from the University of Padova (Progetti di ricerca di Ateneo – CPDA114940) to F.P. S.M. was supported by a Fondazione Umberto Veronesi Fellowship.
Author contributions
S.C.N. conceived and designed the study, performed research, analyzed data and wrote the manuscript. M.M., M.P., E.M., M.C., L.Q.T., and S.M. helped with experiments, partly analyzed data and revised the manuscript; R.Z., F.A., A.V., and G.B. contributed with patients’ samples; L.T. and G.S. contributed with patients’ samples and provided funding; A.N. provided funding and critical advices during elaboration of data. F.P. supported the design of the study, provided funding, supervised research and data analysis, and revised the manuscript.
Conflict of interest
The authors declare that they have no conflict of interest.
Footnotes
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Supplementary Information accompanies this paper at 10.1038/s41408-018-0053-z.
References
- 1.Anderson KC, Carrasco RD. Pathogenesis of myeloma. Annu. Rev. Pathol. 2011;6:249–274. doi: 10.1146/annurev-pathol-011110-130249. [DOI] [PubMed] [Google Scholar]
- 2.Morgan GJ, Walker Ba, Davies FE. The genetic architecture of multiple myeloma. Nat. Rev. Cancer. 2012;12:335–348. doi: 10.1038/nrc3257. [DOI] [PubMed] [Google Scholar]
- 3.Landgren O, et al. Monoclonal gammopathy of undetermined significance (MGUS) consistently precedes multiple myeloma: a prospective study. Blood. 2009;113:5412–5417. doi: 10.1182/blood-2008-12-194241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Noonan K, Borrello I. The immune microenvironment of myeloma. Cancer Microenviron. 2011;4:313–323. doi: 10.1007/s12307-011-0086-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Vetter IR, Wittinghofer A. The guanine nucleotide-binding switch in three dimensions. Science. 2001;294:1299–1304. doi: 10.1126/science.1062023. [DOI] [PubMed] [Google Scholar]
- 6.Aspenström P, Ruusala A, Pacholsky D. Taking Rho GTPases to the next level: the cellular functions of atypical Rho GTPases. Exp. Cell Res. 2007;313:3673–3679. doi: 10.1016/j.yexcr.2007.07.022. [DOI] [PubMed] [Google Scholar]
- 7.Vega FM, Ridley AJ. Rho GTPases in cancer cell biology. FEBS Lett. 2008;582:2093–2101. doi: 10.1016/j.febslet.2008.04.039. [DOI] [PubMed] [Google Scholar]
- 8.Raftopoulou M, Hall A. Cell migration: Rho GTPases lead the way. Dev. Biol. 2004;265:23–32. doi: 10.1016/j.ydbio.2003.06.003. [DOI] [PubMed] [Google Scholar]
- 9.Risse SL, et al. SH3-mediated targeting of Wrch1/RhoU by multiple adaptor proteins. Biol. Chem. 2013;394:421–432. doi: 10.1515/hsz-2012-0246. [DOI] [PubMed] [Google Scholar]
- 10.Dickover M, et al. The atypical Rho GTPase, RhoU, regulates cell-adhesion molecules during cardiac morphogenesis. Dev. Biol. 2014;389:182–191. doi: 10.1016/j.ydbio.2014.02.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Schiavone D, et al. The RhoU/Wrch1 Rho GTPase gene is a common transcriptional target of both the gp130/STAT3 and Wnt-1 pathways. Biochem. J. 2009;421:283–292. doi: 10.1042/BJ20090061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Fort P, et al. Activity of the RhoU/Wrch1 GTPase is critical for cranial neural crest cell migration. Dev. Biol. 2011;350:451–463. doi: 10.1016/j.ydbio.2010.12.011. [DOI] [PubMed] [Google Scholar]
- 13.Todoerti K, et al. Transcriptional characterization of a prospective series of primary plasma cell leukemia revealed signatures associated with tumor progression and poorer outcome. Clin. Cancer Res. 2013;19:3247–3258. doi: 10.1158/1078-0432.CCR-12-3461. [DOI] [PubMed] [Google Scholar]
- 14.Ronchetti D, et al. Distinct lncRNA transcriptional fingerprints characterize progressive stages of multiple myeloma. Oncotarget. 2016;7:14814–14830. doi: 10.18632/oncotarget.7442. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.López-Corral L, et al. Transcriptome analysis reveals molecular profiles associated with evolving steps of monoclonal gammopathies. Haematologica. 2014;99:1365–1372. doi: 10.3324/haematol.2013.087809. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Hideshima T, Bergsagel PL, Kuehl WM, Anderson KC. Advances in biology of multiple myeloma: clinical applications. Blood. 2004;104:607–618. doi: 10.1182/blood-2004-01-0037. [DOI] [PubMed] [Google Scholar]
- 17.Agnelli L, et al. Molecular classification of multiple myeloma: a distinct transcriptional profile characterizes patients expressing CCND1 and negative for 14q32 translocations. J. Clin. Oncol. 2005;23:7296–7306. doi: 10.1200/JCO.2005.01.3870. [DOI] [PubMed] [Google Scholar]
- 18.Agnelli L, et al. Upregulation of translational machinery and distinct genetic subgroups characterise hyperdiploidy in multiple myeloma. Br. J. Haematol. 2007;136:565–573. doi: 10.1111/j.1365-2141.2006.06467.x. [DOI] [PubMed] [Google Scholar]
- 19.Manni, S. et al. Inactivation of CK1α in multiple myeloma empowers drug cytotoxicity by affecting AKT and β-catenin survival signaling pathways. Oncotarget 2017. doi:10.18632/oncotarget.14654. [DOI] [PMC free article] [PubMed]
- 20.Zhan F, et al. The molecular classification of multiple myeloma. Blood. 2006;108:2020–2028. doi: 10.1182/blood-2005-11-013458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Chen L, et al. Identification of early growth response protein 1 (EGR-1) as a novel target for JUN-induced apoptosis in multiple myeloma. Blood. 2010;115:61–70. doi: 10.1182/blood-2009-03-210526. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Hanamura I, Huang Y, Zhan F, Barlogie B, Shaughnessy J. Prognostic value of cyclin D2 mRNA expression in newly diagnosed multiple myeloma treated with high-dose chemotherapy and tandem autologous stem cell transplantations. Leukemia. 2006;20:1288–1290. doi: 10.1038/sj.leu.2404253. [DOI] [PubMed] [Google Scholar]
- 23.Heuck CJ, et al. Five gene probes carry most of the discriminatory power of the 70-gene risk model in multiple myeloma. Leukemia. 2014;28:2410–2413. doi: 10.1038/leu.2014.232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Li D, Das S, Yamada T, Samuels HH. The NRIF3 family of transcriptional coregulators induces rapid and profound apoptosis in breast cancer cells. Mol. Cell Biol. 2004;24:3838–3848. doi: 10.1128/MCB.24.9.3838-3848.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Orjalo AV, et al. The Nup107-160 Nucleoporin complex is required for correct bipolar spindle assembly. Mol. Biol. Cell. 2006;17:3806–3818. doi: 10.1091/mbc.E05-11-1061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Platani M, et al. The Nup107-160 nucleoporin complex promotes mitotic events via control of the localization state of the chromosome passenger complex. Mol. Biol. Cell. 2009;20:5260–5275. doi: 10.1091/mbc.E09-05-0377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Thomas Y, Coux O, Baldin V. βTrCP-dependent degradation of CDC25B phosphatase at the metaphase-anaphase transition is a pre-requisite for correct mitotic exit. Cell Cycle. 2010;9:4338–4350. doi: 10.4161/cc.9.21.13593. [DOI] [PubMed] [Google Scholar]
- 28.Osman F, Whitby MC. Emerging roles for centromere-associated proteins in DNA repair and genetic recombination. Biochem. Soc. Trans. 2013;41:1726–1730. doi: 10.1042/BST20130200. [DOI] [PubMed] [Google Scholar]
- 29.Kim H, Fonseca C, Stumpff J. A unique kinesin-8 surface loop provides specificity for chromosome alignment. Mol. Biol. Cell. 2014;25:3319–3329. doi: 10.1091/mbc.E14-06-1132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Song H, Park JE, Jang CY. DDA3 targets Cep290 into the centrosome to regulate spindle positioning. Biochem. Biophys. Res. Commun. 2015;463:88–94. doi: 10.1016/j.bbrc.2015.05.028. [DOI] [PubMed] [Google Scholar]
- 31.Gozuacik D, et al. Identification and functional characterization of a new member of the human Mcm protein family: hMcm8. Nucleic Acids Res. 2003;31:570–579. doi: 10.1093/nar/gkg136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Pollok S, Stoepel J, Bauerschmidt C, Kremmer E, Nasheuer HP. Regulation of eukaryotic DNA replication at the initiation step. Biochem. Soc. Trans. 2003;31:266–269. doi: 10.1042/bst0310266. [DOI] [PubMed] [Google Scholar]
- 33.Henninger EE, Pursell ZF, Henninger EE, Pursell ZF. DNA polymerase ε and its roles in genome stability. Iubmb. Life. 2014;66:339–351. doi: 10.1002/iub.1276. [DOI] [PubMed] [Google Scholar]
- 34.Wäsch R, Robbins Ja, Cross FR. The emerging role of APC/CCdh1 in controlling differentiation, genomic stability and tumor suppression. Oncogene. 2010;29:1–10. doi: 10.1038/onc.2009.325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Hurt EM, et al. Overexpression of c-maf is a frequent oncogenic event in multiple myeloma that promotes proliferation and pathological interactions with bone marrow stroma. Cancer Cell. 2004;5:191–199. doi: 10.1016/S1535-6108(04)00019-4. [DOI] [PubMed] [Google Scholar]
- 36.Boncela J, et al. Plasminogen activator inhibitor type 1 interacts with alpha3 subunit of proteasome and modulates its activity. J. Biol. Chem. 2011;286:6820–6831. doi: 10.1074/jbc.M110.173781. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Bakkenist CJ, Kastan MB. DNA damage activates ATM through intermolecular autophosphorylation and dimer dissociation. Nature. 2003;421:499–506. doi: 10.1038/nature01368. [DOI] [PubMed] [Google Scholar]
- 38.Jeong SJ, et al. Role of RNA polymerase II carboxy terminal domain phosphorylation in DNA damage response. J. Microbiol. 2005;43:516–522. [PubMed] [Google Scholar]
- 39.Klungland A, Bjelland S. Oxidative damage to purines in DNA: role of mammalian Ogg1. DNA Repair. 2007;6:481–488. doi: 10.1016/j.dnarep.2006.10.012. [DOI] [PubMed] [Google Scholar]
- 40.Wu J, Lu LY, Yu X. The role of BRCA1 in DNA damage response. Protein Cell. 2010;1:117–123. doi: 10.1007/s13238-010-0010-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Roy N, Bagchi S, Raychaudhuri P. Damaged DNA binding protein 2 in reactive oxygen species (ROS) regulation and premature senescence. Int. J. Mol. Sci. 2012;13:11012–11026. doi: 10.3390/ijms130911012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Eddy S, et al. Human Rev1 polymerase disrupts G-quadruplex DNA. Nucleic Acids Res. Nucleic Acids Res. 2014;42:3272–3285. doi: 10.1093/nar/gkt1314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Abdou I, Poirier GC, Hendzel MJ, Weinfeld M. DNA ligase III acts as a DNA strand break sensor in the cellular orchestration of DNA strand break repair. Nucleic Acids Res. 2015;43:875–892. doi: 10.1093/nar/gku1307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Saito Y, Komatsu K. Functional role of NBS1 in radiation damage response and translesion DNA synthesis. Biomolecules. 2015;5:1990–2002. doi: 10.3390/biom5031990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Lin H, et al. An activated JAK/STAT3 pathway and CD45 expression are associated with sensitivity to Hsp90 inhibitors in multiple myeloma. Exp. Cell Res. 2013;319:600–611. doi: 10.1016/j.yexcr.2012.12.006. [DOI] [PubMed] [Google Scholar]
- 46.Mahmoud MS, Ishikawa H, Fujii R, Kawano MM. Induction of CD45 expression and proliferation in U-266 myeloma cell line by interleukin-6. Blood. 1998;92:3887–3897. [PubMed] [Google Scholar]
- 47.Faure S, Fort P. Atypical RhoV and RhoU GTPases control development of the neural crest. Small GTPases. 2011;2:310–313. doi: 10.4161/sgtp.18086. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Schust J, Sperl B, Hollis A, Mayer TU, Berg T. Stattic: a small-molecule inhibitor of STAT3 activation and dimerization. Chem. Biol. 2006;13:1235–1242. doi: 10.1016/j.chembiol.2006.09.018. [DOI] [PubMed] [Google Scholar]
- 49.Xu Y, et al. Immunomodulatory drugs reorganize cytoskeleton by modulating Rho GTPases. Blood. 2009;114:338–345. doi: 10.1182/blood-2009-02-200543. [DOI] [PubMed] [Google Scholar]
- 50.Zhu YX, et al. Cereblon expression is required for the antimyeloma activity of lenalidomide and pomalidomide. Blood. 2011;118:4771–4779. doi: 10.1182/blood-2011-05-356063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Escoubet-Lozach L, et al. Pomalidomide and lenalidomide induce p21WAF-1 expression in both lymphoma and multiple myeloma through a LSD1-mediated epigenetic mechanism. Cancer Res. 2009;69:7347–7356. doi: 10.1158/0008-5472.CAN-08-4898. [DOI] [PubMed] [Google Scholar]
- 52.Podar K, Chauhan D, Anderson KC. Bone marrow microenvironment and the identification of new targets for myeloma therapy. Leukemia. 2009;23:10–24. doi: 10.1038/leu.2008.259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Tao W, Pennica D, Xu L, Kalejta RF, Levine AJ. Wrch-1, a novel member of the Rho gene family that is regulated by Wnt-1. Genes Dev. 2001;15:1796–1807. doi: 10.1101/gad.894301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Chuang Y, Valster A, Coniglio SJ, Backer JM, Symons M. The atypical Rho family GTPase Wrch-1 regulates focal adhesion formation and cell migration. J. Cell Sci. 2007;120:1927–1934. doi: 10.1242/jcs.03456. [DOI] [PubMed] [Google Scholar]
- 55.Quach H, Kalff A, Spencer A. Lenalidomide in multiple myeloma: current status and future potential. Am. J. Hematol. 2012;87:1089–1095. doi: 10.1002/ajh.23234. [DOI] [PubMed] [Google Scholar]
- 56.Fink EC, Ebert BL. The novel mechanism of lenalidomide activity. Blood. 2015;126:2366–2369. doi: 10.1182/blood-2015-07-567958. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Roboz GJ. Novel approaches to the treatment of acute myeloid leukemia. Hematol. Am. Soc. Hematol. Educ. Program. 2011;2011:43–50. doi: 10.1182/asheducation-2011.1.43. [DOI] [PubMed] [Google Scholar]
- 58.Kotla V, et al. Mechanism of action of lenalidomide in hematological malignancies. J. Hematol. Oncol. 2009;2:36. doi: 10.1186/1756-8722-2-36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Tubi LQ, et al. Protein kinase CK2 regulates AKT, NF-κB and STAT3 activation, stem cell viability and proliferation in acute myeloid leukemia. Leukemia. 2016;31:292–300. doi: 10.1038/leu.2016.209. [DOI] [PubMed] [Google Scholar]
- 60.Tusher VG, Tibshirani R, Chu G. Significance analysis of microarrays applied to the ionizing radiation response. Proc. Natl Acad. Sci. USA. 2001;98:5116–5121. doi: 10.1073/pnas.091062498. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.